Heart ACS
The HarEfield Acute MyocaRdial InfarcTion Cohort
(Heart-ACS)
Miles Dalby, Vas Panoulas, Tito Kabir, Richard Grocott-Mason, Allan Davies, Paula Rogers, Geraldine Sloanes, Jackie Donovan and Thomas F. Lüscher
Royal Brompton and Harefield Hospital NHS Foundation Trust
London and Harefield, U.K.

About
Background
Acute coronary syndromes (ACS), i.e. patients presenting with ST-segment elevation myocardial infarction (STEMI), non ST-segment elevation myocardial infarction (non-STEMI) or unstable angina, are still a major cause of morbidity and mortality in the United Kingdom and beyond. In spite of the enormous progress made in the last decades, the in-hospital mortality has plateaued recently (Fig. 1) and the event rate after the infarction is still high with one in 8 patients having a second event (i.e. death, myocardial infarction, heart failure, revascularization) within a year of follow-up.
After the acute event, risk stratification is important and will become even more sophisticated than it currently is with the advent of anatomic risk scores (SYNTAX II Score), novel biomarkers and novel drugs allowing for more precise characterisation of the patients individual risk and tailored secondary prevention strategies (Precision Medicine). Indeed, particularly the upcoming treatment options with biologicals (i.e. monoclonal antibodies, e.g. against PCSK9) and genetic tools (i.e. RNA interference, antisense technology) will require precise risk assessment for cost-effective use of these promising new tools.
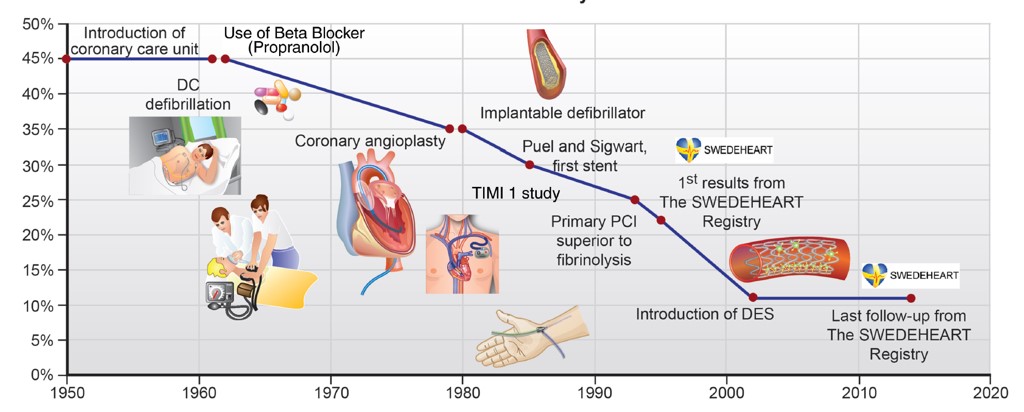
Figure 1: Development of in-hospital mortality over the last decades in patients presenting with myocardial infarction. (Lüscher T.F. and Obeid S. Eur. Heart J. 2018)
Plan
Scientific Plan
We plan to establish a prospective ACS registry (Heart-ACS) at the Harefield Hospital enrolling all patients presenting with ST-segment elevation myocardial infarction (STEMI), non ST-segment elevation myocardial infarction (non-STEMI), unstable angina, Tako Tsubo syndrome or coronary dissection as defined by the current ESC Guidelines and Consensus Papers, irrespective of their management (Figure 2). Patients will be documented during the hospital stay and seen at the institution at the post-pPCI clinic at 6 months. A telephone call will be done at 12 months.
To that end, we have established a eCRF (electronic case report form) encompassing all relevant data (clinical, anatomic, clinical chemistry and biomarkers etc.) during the hospital stay and at one year follow-up with an independent events adjudication committee consisting of three experienced cardiologists assuring for high quality data.

Furthermore, we will collect whole blood (for DNA analysis), serum and plasma samp-les and store them at -80oC in a specially assigned freezer. Samples may be collected for future unspecified research and consent will be obtained to store residual samples within the existing Cardiovascular Research Centre Biobank at RBHT.
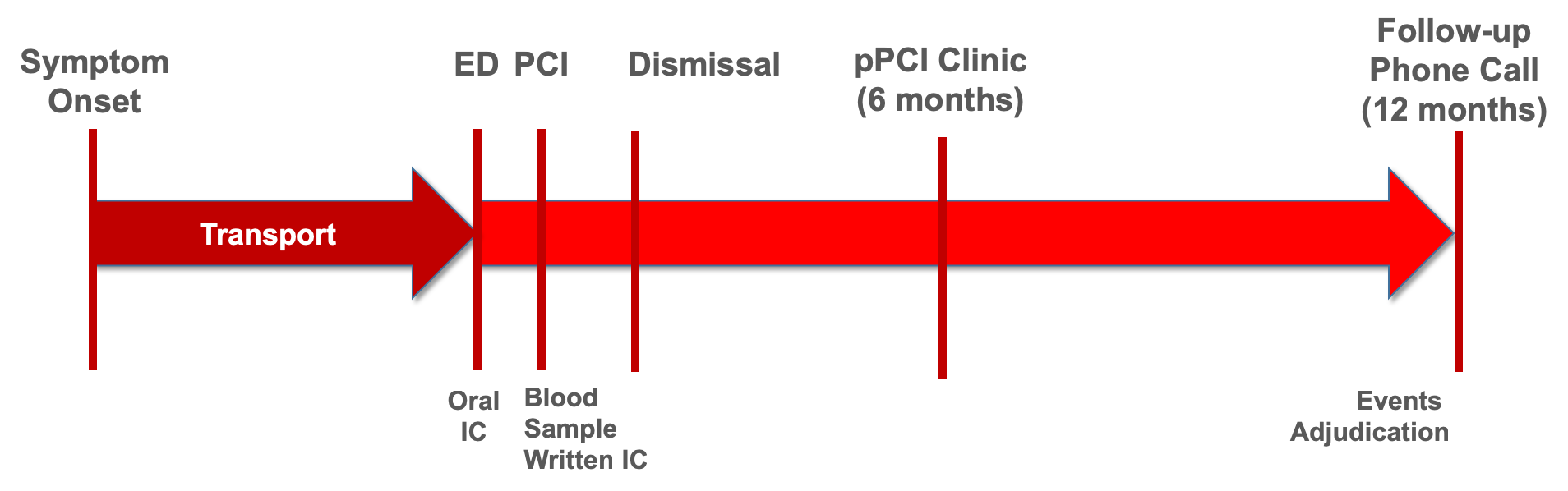
Figure 2: Outline of the Heart-ACS Cohort at Harefield Hospital. ED = Emergency Department; IC = informed consent; pPCI = primary percutaneous coronary intervention.
Scientific opportunities
Such a database will be an enormous scientific and academic opportuntiy for fellows and staff at Harefield alike. Not only will this allow for the evaluation of novel biomarkers and outcomes studies, but also clinial trials could be easily nested within the registry.

Organisational Structure
1. Steering Committee
Miles Dalby (Primary Investigator), Vas Panoulas, Tito Kabir, Richard Grocott-Mason, Allan Davies, Paula Rogers, Geraldine Sloanes, Jackie Donovan and Thomas F. Lüscher (Senior Investigator)
4. Biobank
A biobank (Manager: Geraldine Sloanes) with -80oC refrigerators and a tissue/blood samples tracking system equipped with barcodes is available at the Harefield campus. During office hours, samples will be collected from the research nurse team and documented through the LabVantage system. -80o C freezer space will be secured in the Heart Science Centre. During nights and weekends, the interventional nursing staff will be responsible for transport of the blood samples to the core lab.
2. Events Adjudication Committee
Three experienced cardiologists from outside the institution or unrelated to the project from inside the institution.
5. Research Nursing Staff
The research nurses (Head: Paula Rogers) are highly motivated to help with this project and received some extra personal resources for this project.
3. Core Lab
This will the Department of Clinical Biochemistry (Director: Jackie Donovan). They are staffed 24/7 and are willing to accept the blood samples, centrifuge them and store them into a series of Eppendorf tubes for later storage.
6. Interventional Nurses
The interventional nurses will collect the blood samples retrieved from the sheet by the operator in charge and store them on ice (4oC). During working hours the research nurse team will further handle the samples, while at night and on weekends the interventional nurses would need to bring them across the road to the Department of Clinical Biochemistry for processing.
Our Partners

Address for Correspondence

Professor Thomas F. Lüscher, MD, FRCP:

Dr Miles Dalby
Consultant Cardiologist
Honorary Senior Lecturer Imperial College
Royal Brompton & Harefield Hospitals
Heart Division
London, UK